SiteVault What's New in 26R1
Release Date: April 17, 2025
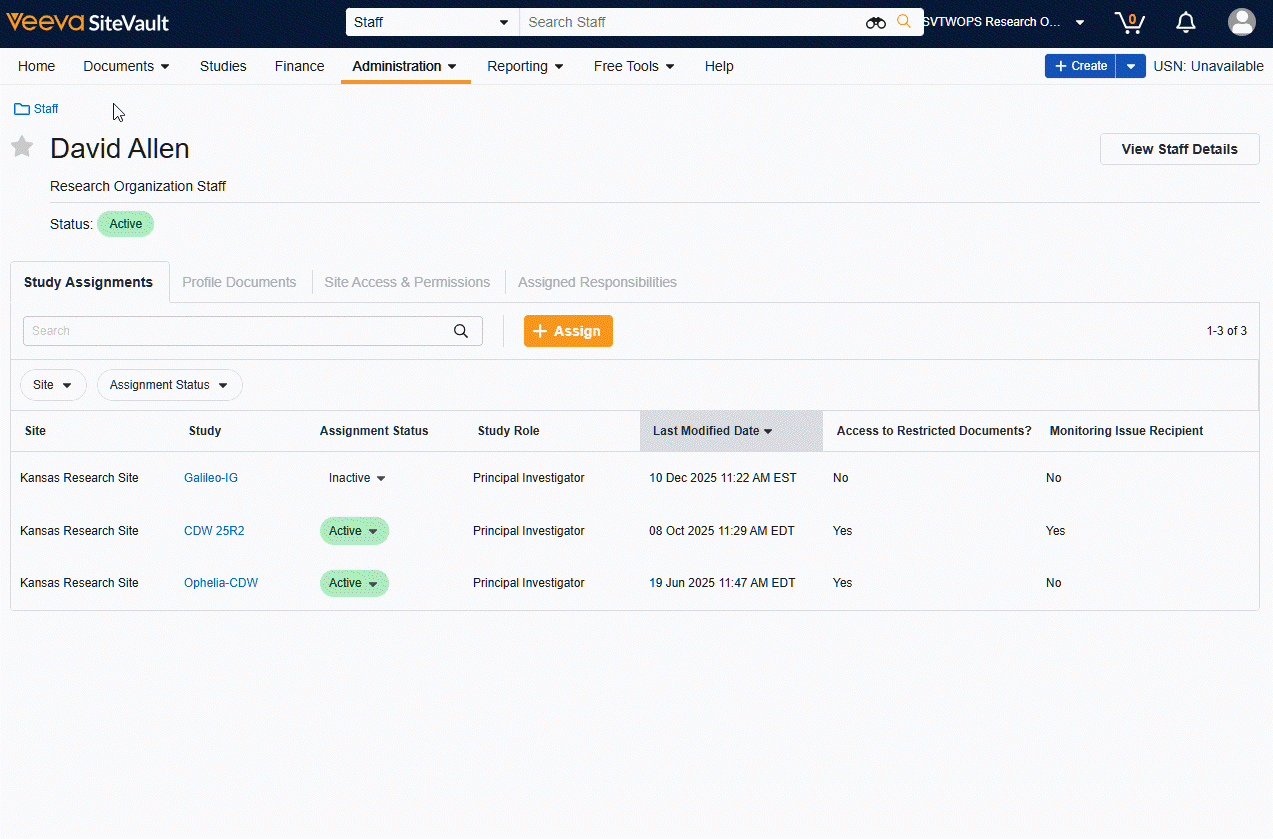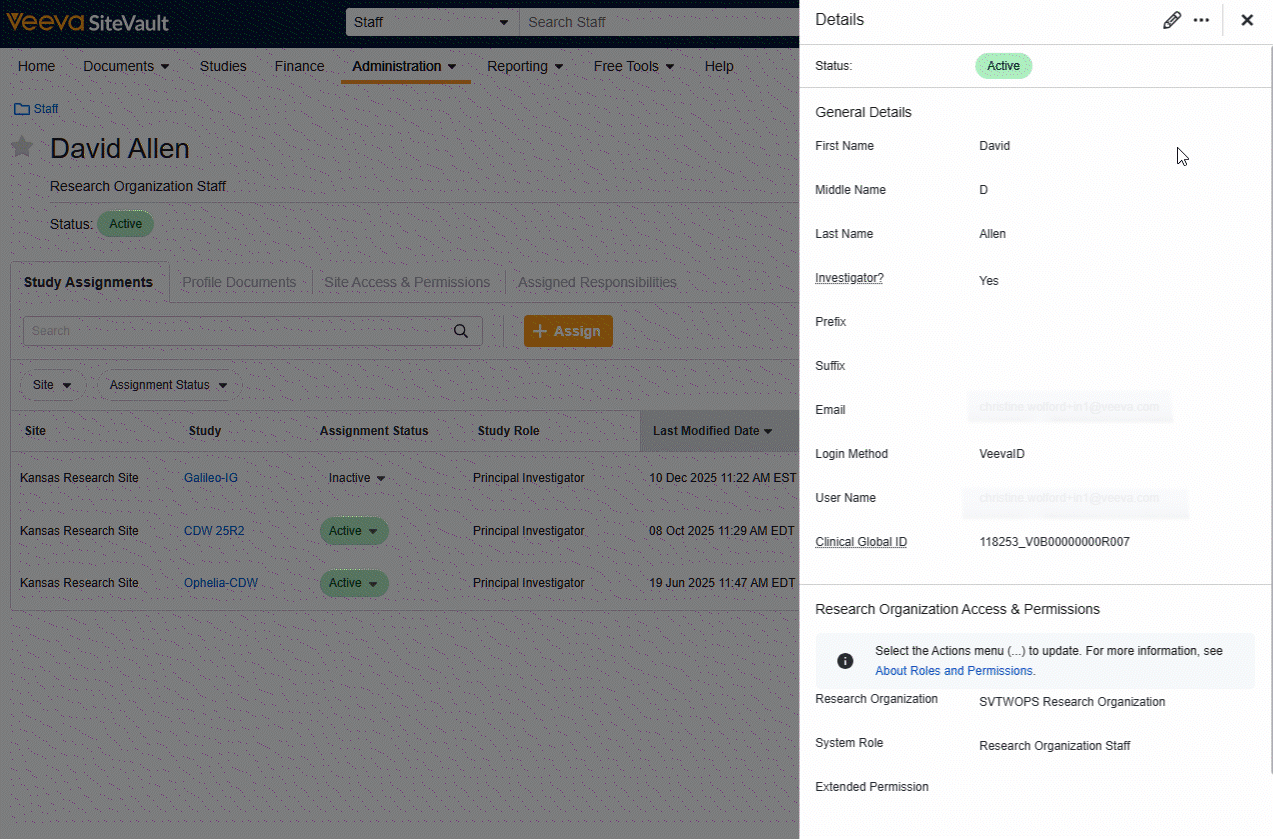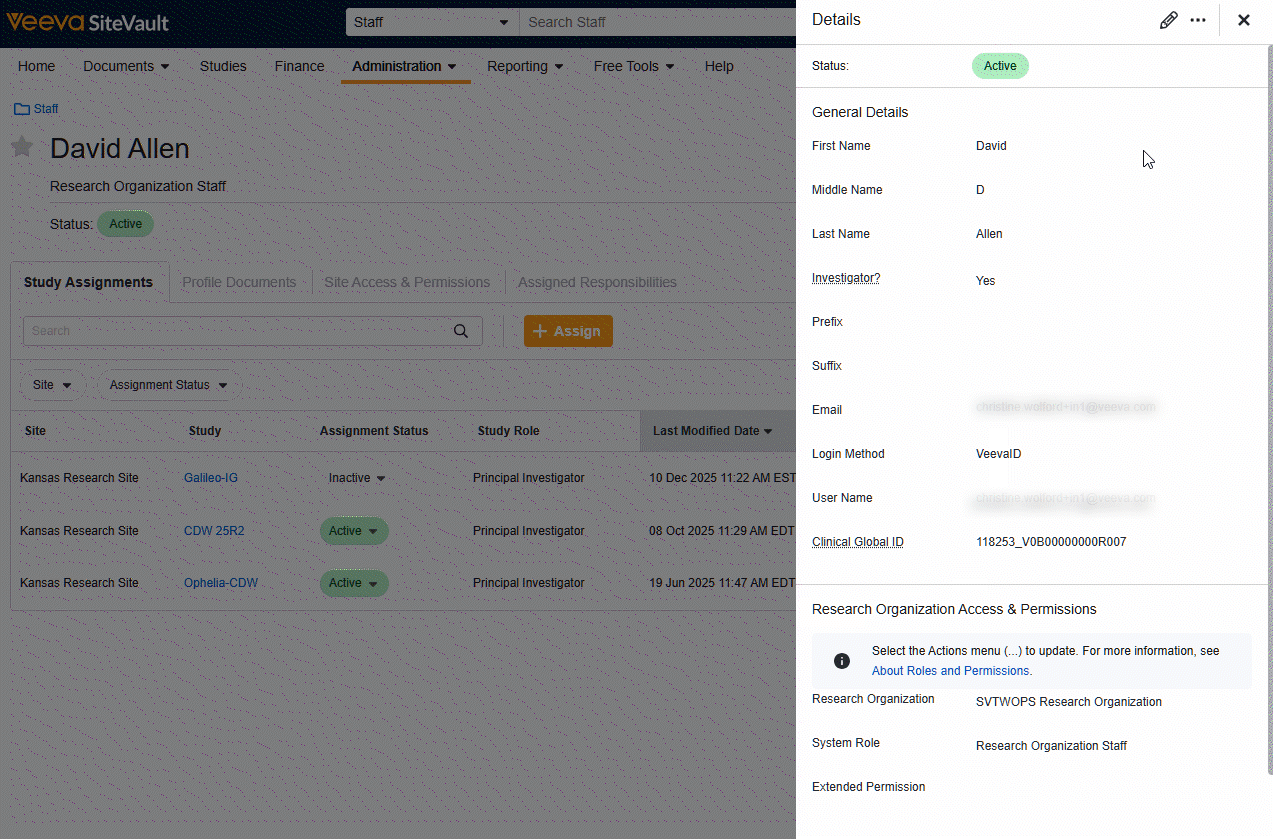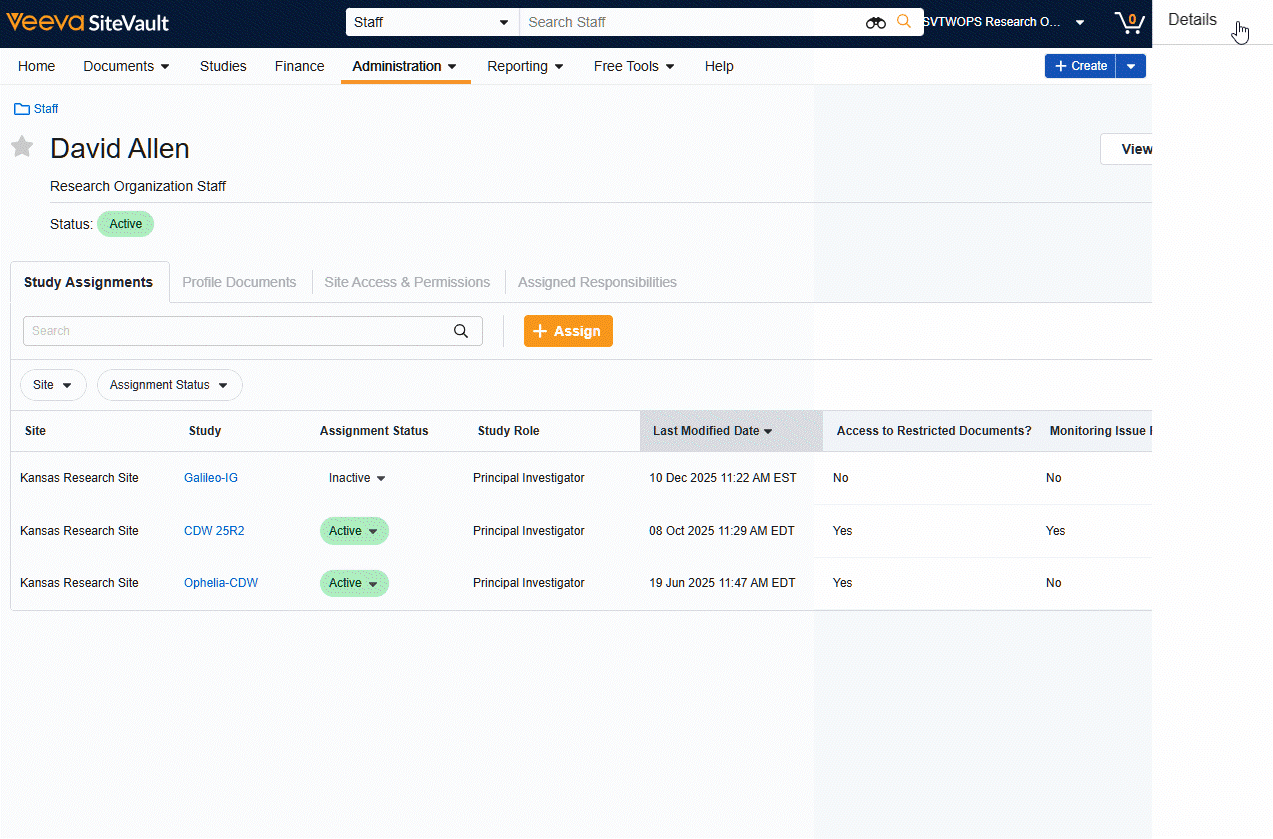
Site Staff and External User Pages Refresh
Users will find a streamlined workflow and redesigned interface for Site Staff and Monitor/External User pages. The tab layout offers speedy navigation to information and tools related to individual users and the View Details button offers simplified actions for updating user general information and research organization permissions.
Digital Financial Disclosure Forms
This feature allows site staff to initiate a digital Financial Disclosure Form workflow for one or many investigators simultaneously. Investigators receive a task to respond to questions and then e-sign the completed form, all within SiteVault. The process renders a completed standard Financial Disclosure Form document for each investigator and maintains document history through version stacking. The ability to upload a Financial Disclosure Form is still available.
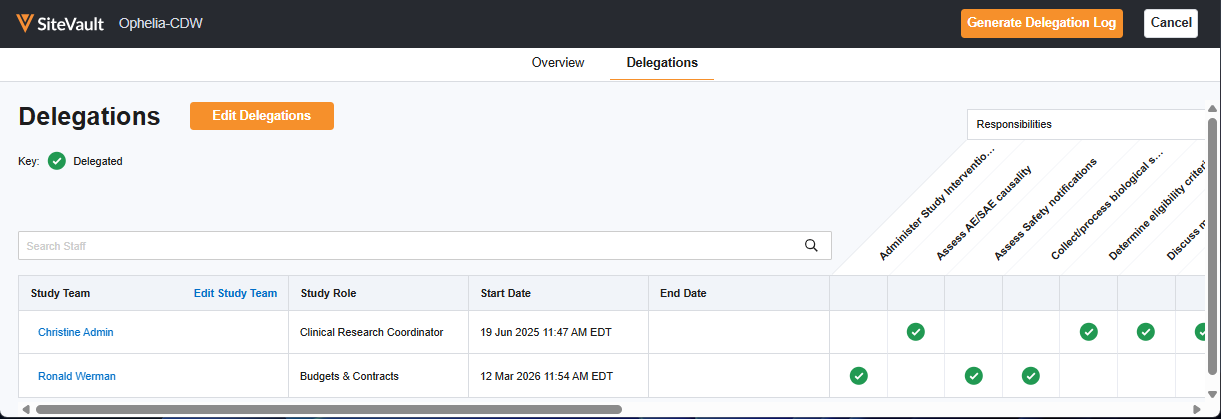
Redesigned Digital Delegation Log
We are pleased to introduce a refreshed Digital Delegation experience, offering a significant update designed to provide greater flexibility and efficiency when managing delegation logs in SiteVault.
- Manage Delegations: The study’s Team tab now includes a Manage Delegation button which navigates to the Delegations Editor. This is an intuitive workspace for managing staff responsibilities and start/end dates in real-time, providing a clear overview of your study’s delegations at all times.
- Flexible Approvals: Sites now have the option to send DOA logs directly to the PI for signature or include a staff acceptance step, depending on your site's review process.
- Inactive Team Members: You can now manage and correct delegation data for inactive study team members directly, ensuring your logs remain accurate throughout the entire study lifecycle.
- Improved DOA Experience: The updated eDOA output features a clickable Table of Contents for faster navigation and a standardized E-Signature Page for improved compliance documentation.
Current DDL users, for more information, see the Digital Delegation 2.0 Transition Guide.
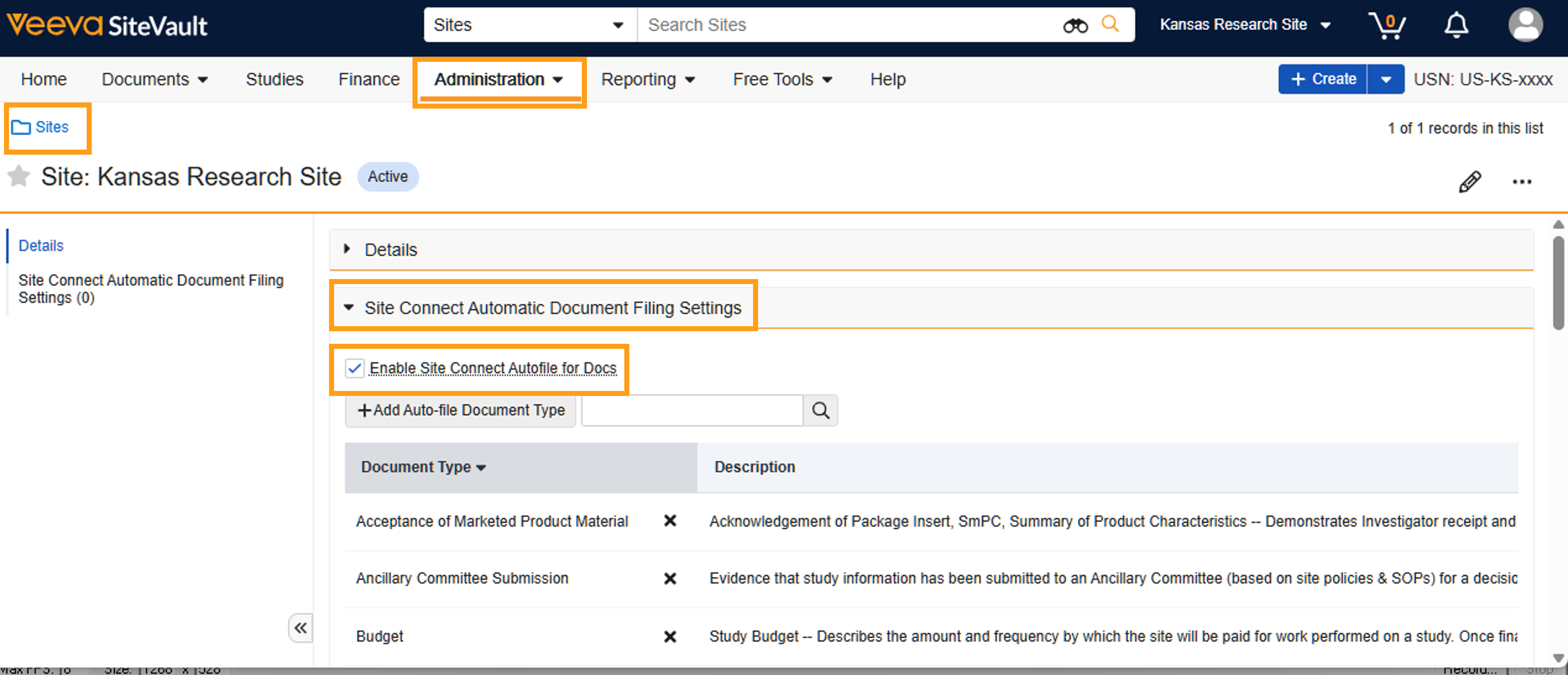
Site Connect Automatic Document Filing
SiteVault allows you to automate the filing of documents from Site Connect’s Document Exchange directly into your Study eBinder. Previously, automatic filing configurations were managed within Site Connect; these settings have now been moved directly into SiteVault. By choosing to automate document filing, you reduce the need for manual downloads, ensuring that important study documents are available in your eISF as soon as they are pushed from your sponsor or CRO's Vault.
Automatic filing settings are managed at the site level (Administration > Sites) and apply to all of your connected studies. However, you can apply an override at the study level if specific studies require a different approach.
Optional Site Signature
To streamline the approval process when a site signature is not needed or appropriate, site staff can approve completed eConsent forms without a countersignature. During a Sign or Reject Document task, selecting the Make Current without Signing option immediately approves the eConsent, bypassing the prompt for site staff credentials and the creation of an electronic signature. For an eConsent to not require a site signature, the Site Member signatory role must not be selected during the eConsent editing process.
Send a Copy of a Consent Form
The Send a Copy action streamlines how sites share signed Informed Consent Forms (ICFs) with participants and their signatories. Sites can provide a signed copy of an ICF to any signatory (with a valid email address on file) via the document's Action menu, eliminating the need to download files and send them through external channels. Signatories receive an email notification providing them access to view the document in the MyVeeva for Patients app.
eConsent Preview Link Expiration
This feature optimizes the eConsent review process by removing expiration limits on preview links for steady state versions, ensuring sponsors have a persistent record of the patient experience. To maintain data integrity, link availability is now strictly tied to the document's presence in SiteVault; if a document is deleted, its preview link is immediately invalidated.
CTMS General Enhancements
The following general CTMS enhancements offer an improved user experience:
- For improved navigation, the Schedule Builder button is added to the Study > Schedules tab.
- On the Budget > Participant Fees page, the Edit Participant Fees button is relabeled to Edit Fee Amounts.
- When creating a billable item from a site fee, the Count column has been updated to Usage.
- When creating a billable item from a site fee, a search bar is provided to easily locate the needed site fee.
- New fields are added to Participant Details for better participant pool evaluation.
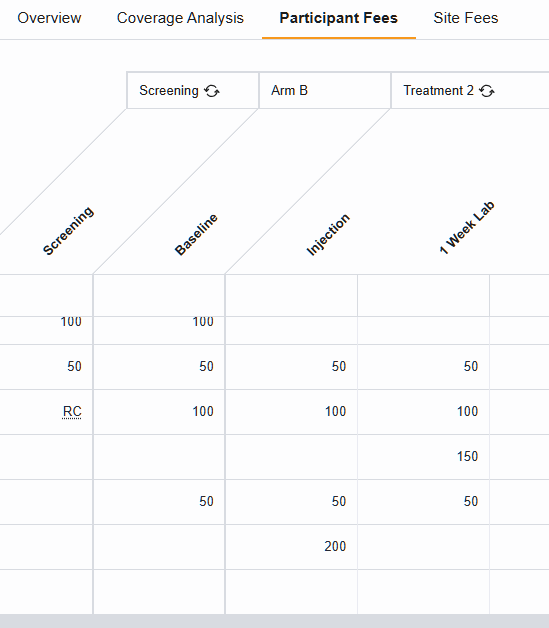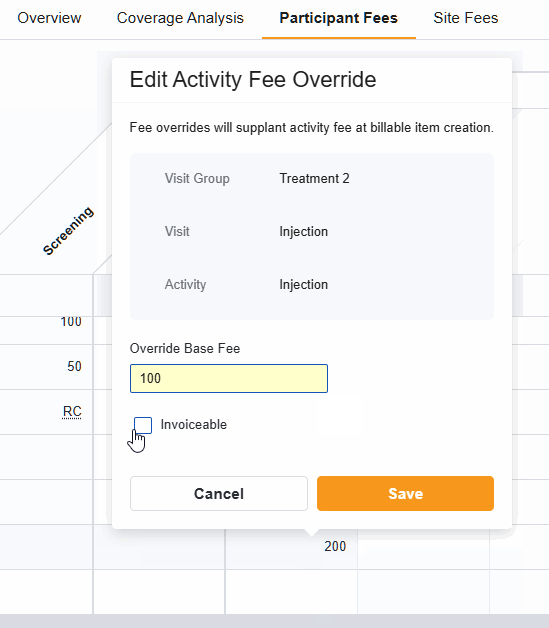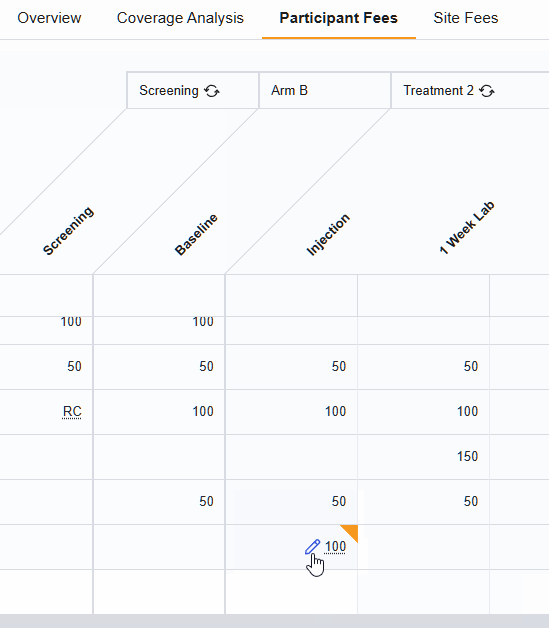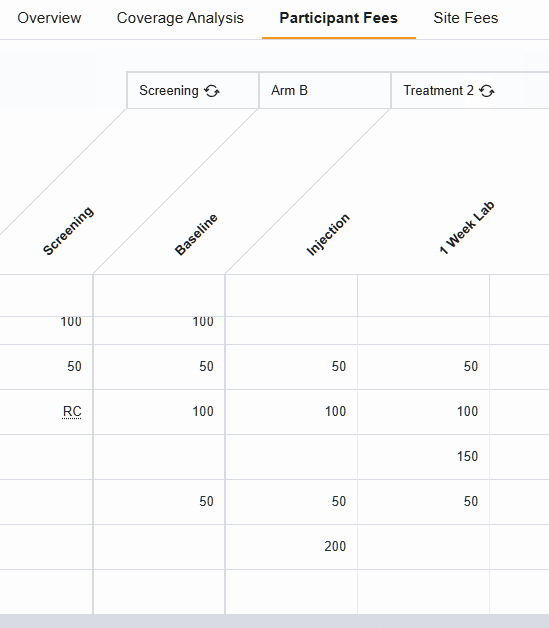
Activity Fee Override
The Activity Fee Override feature gives you the flexibility to deviate from a standard fee schedule for specific, unique situations within a study budget. With this tool, you can change the fee for a specific visit, or change how that activity is invoiced, without having to rewrite the entire budget.
Study Budget: Expense Plan
The Expense Plan (Finance menu) provides a mechanism to capture study operating costs on a per-visit and per-participant basis. This enables you to make informed decisions when negotiating sponsor budgets. Expense plans are version-controlled in the same way as CTMS schedules and budgets. As expenses in the expense plan are incurred upon the completion of participant visits, payable items are automatically generated and tracked on the new Payable Items tab.
The feature provides support for three primary expense categories:
- Activity Expenses: Cost of executing study procedures.
- Personnel Expenses: Hourly cost and effort associated with key study personnel.
- Participant Stipends Cost of participant compensation.
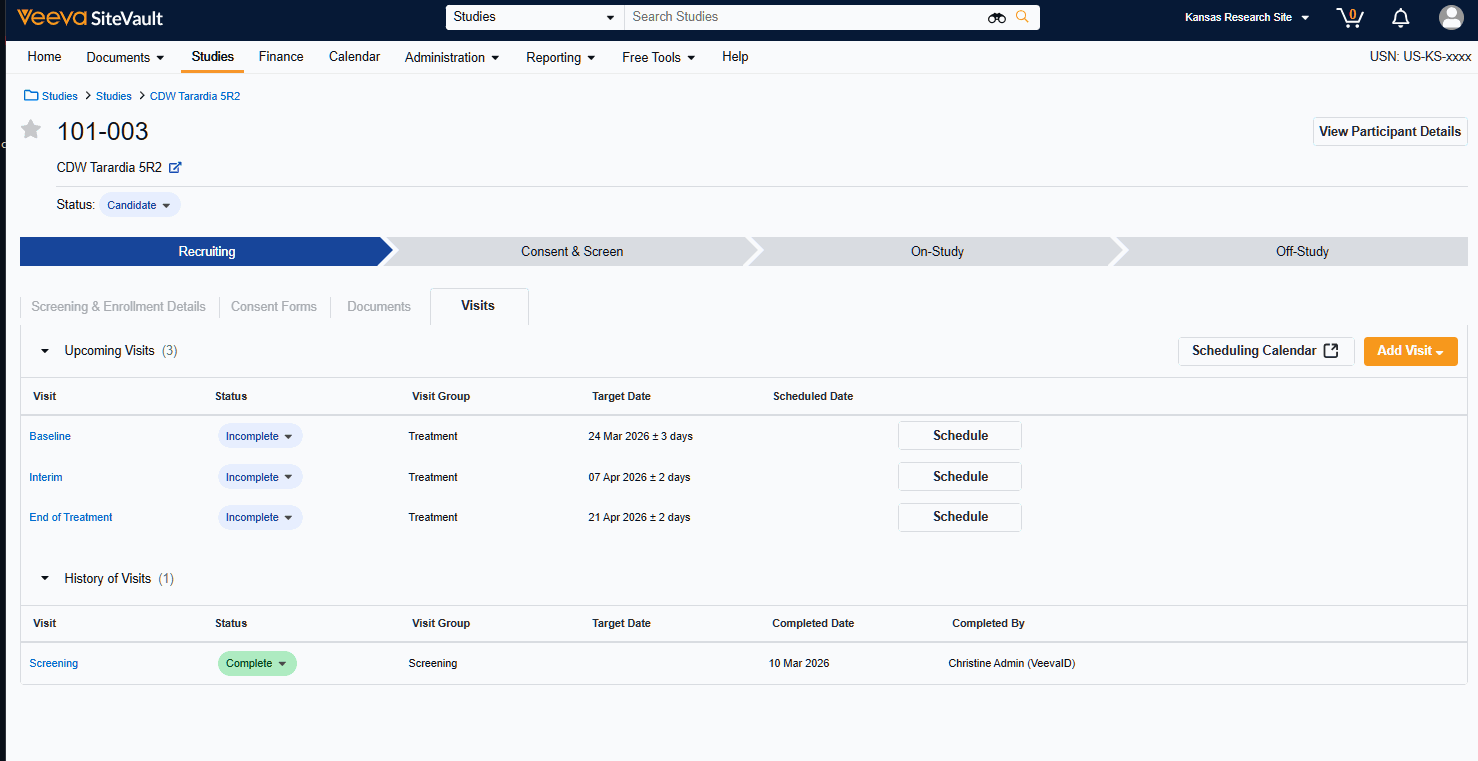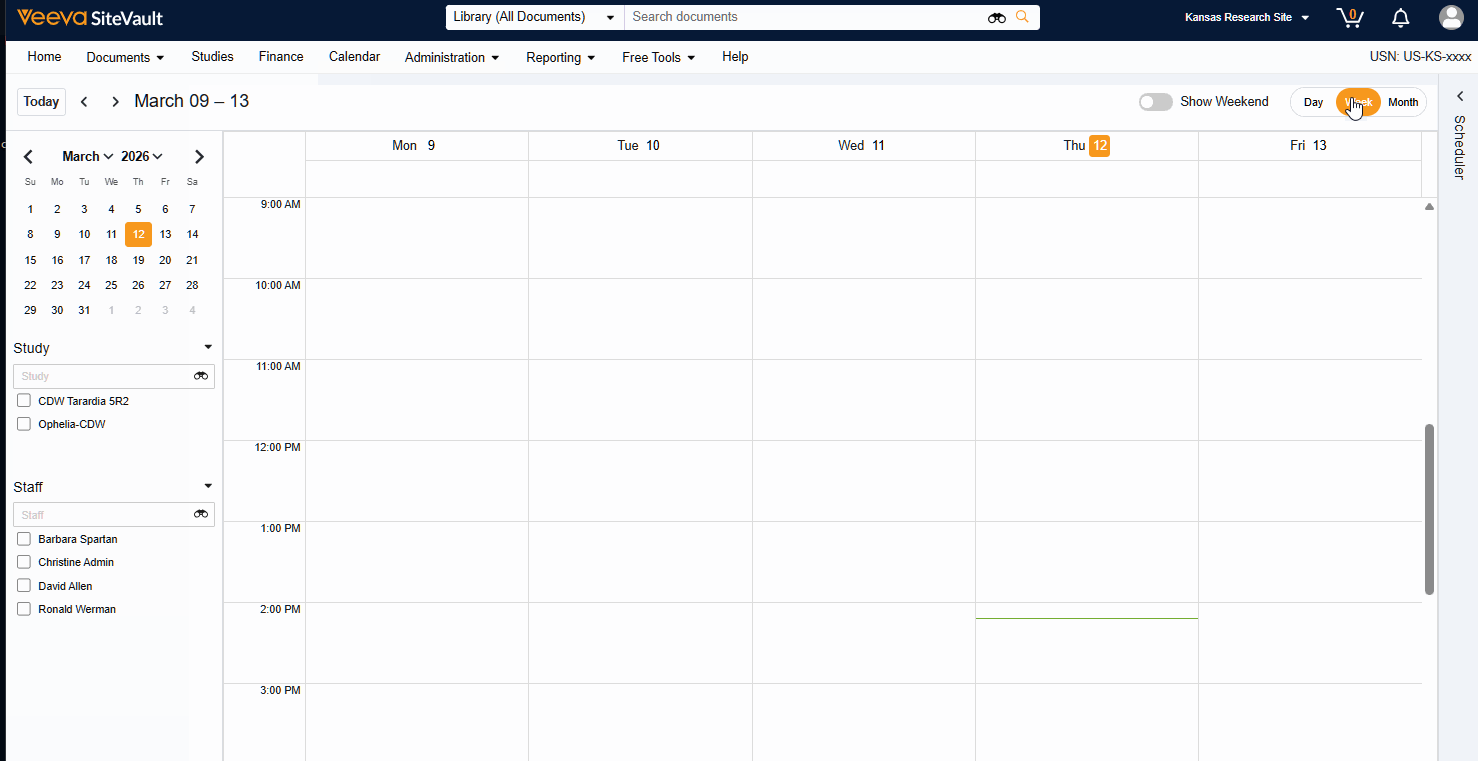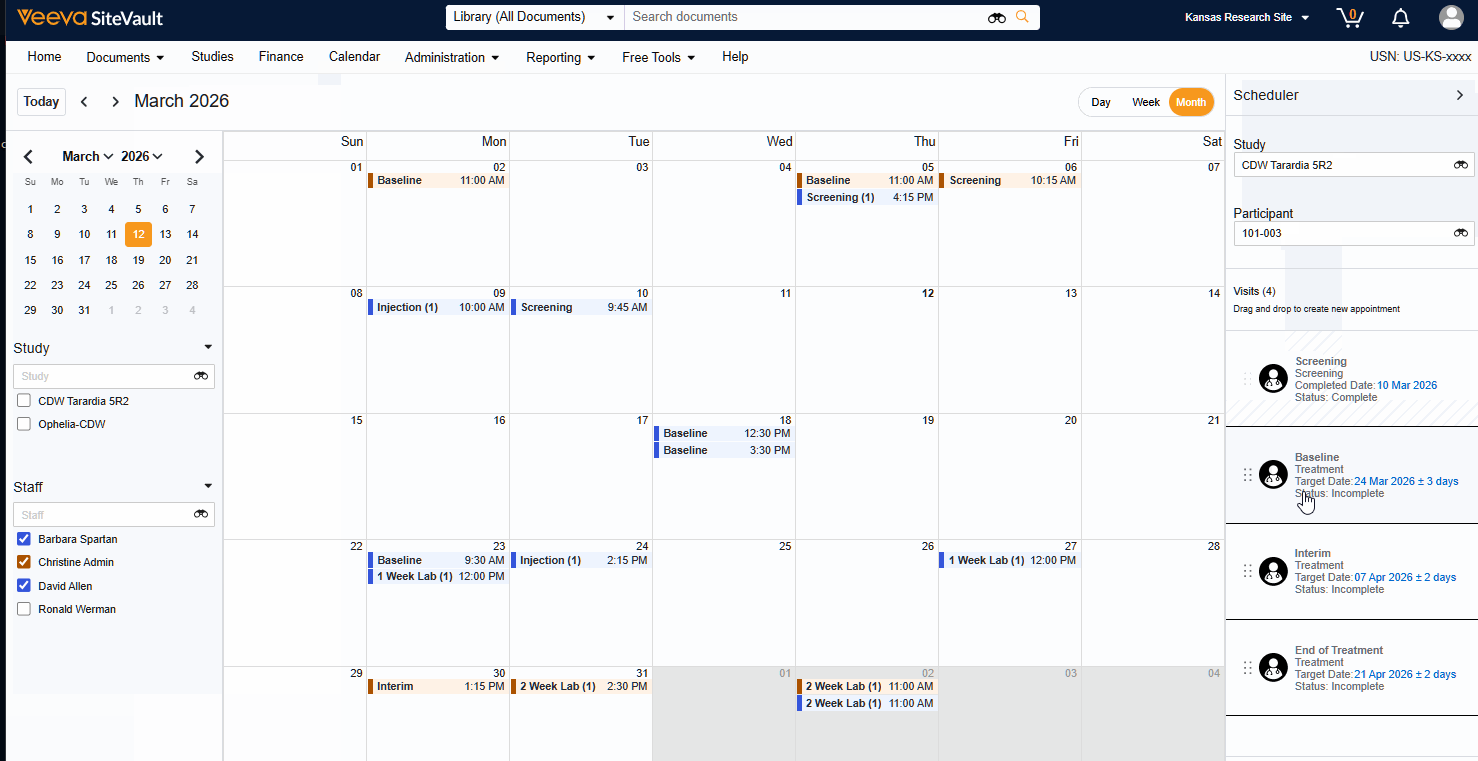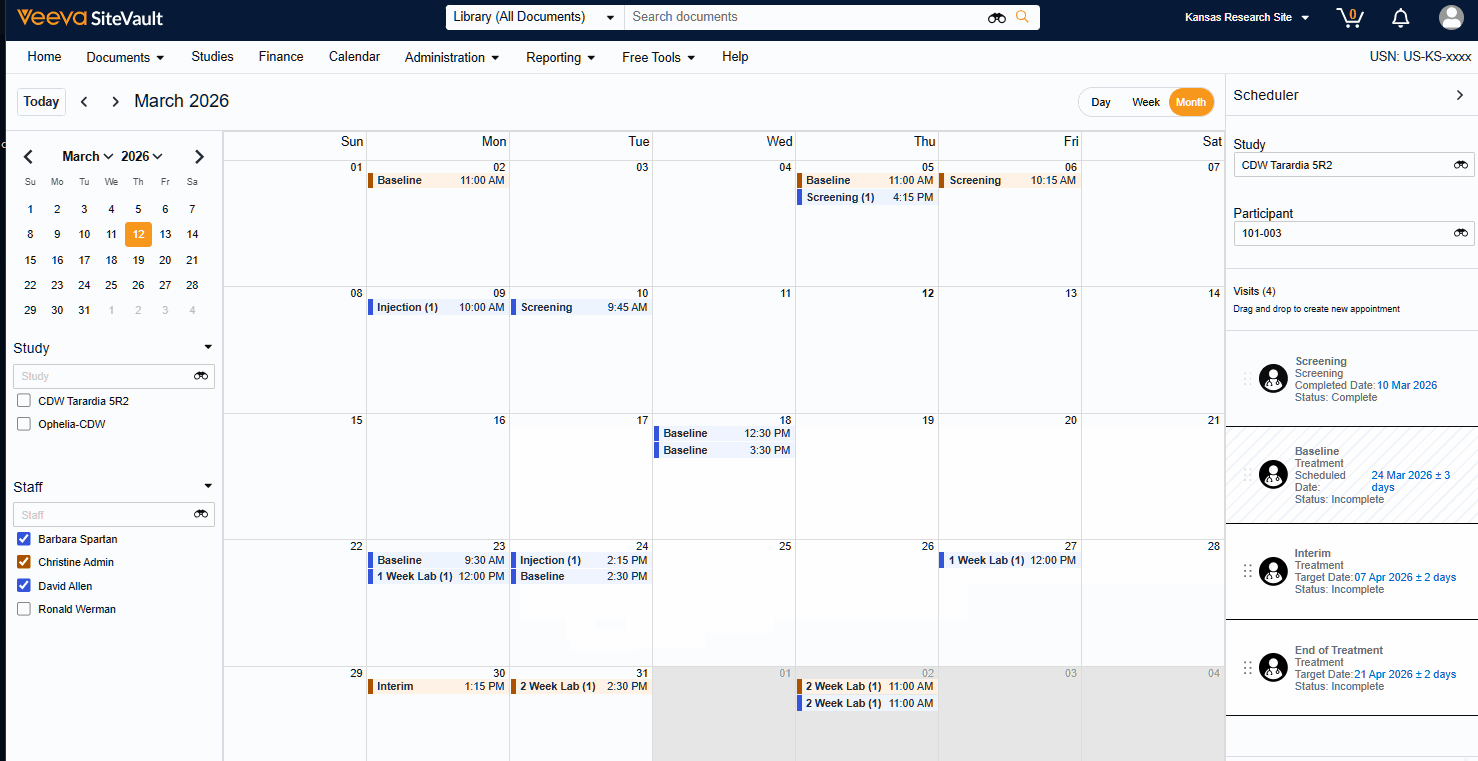
Visit Calendar
The Visit Calendar provides a centralized view of upcoming participant appointments and their associated resources. Designed specifically for sites that use SiteVault CTMS as their system of record, this feature allows you to manage study schedules, staff availability, and participant visits in one location.
- Schedule Participant Visits: Create and manage appointments for specific participant visits at designated dates and times.
- Link Staff to Appointments: Associate specific staff members with scheduled visits to manage workloads.
- Centralized Visibility: View all appointments across all studies in a single, unified calendar.
- Advanced Filtering: Filter the central calendar by Study or Staff to focus on specific scheduling needs
No release notes match the selected filters.