Data Changes
- A data change is required when data entered or generated in the system needs to be modified
- When possible, changes must be made by the user in the Veeva RTSM system
- A Data Change Request (DCR) is necessary when data needs to be modified and the user is unable to modify it in the Veeva RTSM system
- Veeva requires the site to complete a DCR form detailing the requested changes
- All DCRs are managed using the Veeva GSC (help desk)
- Veeva prioritizes any urgent DCRs and may execute prior to obtaining a customer signature if a patient is waiting or the patient’s safety is at risk
Data Change Request Process
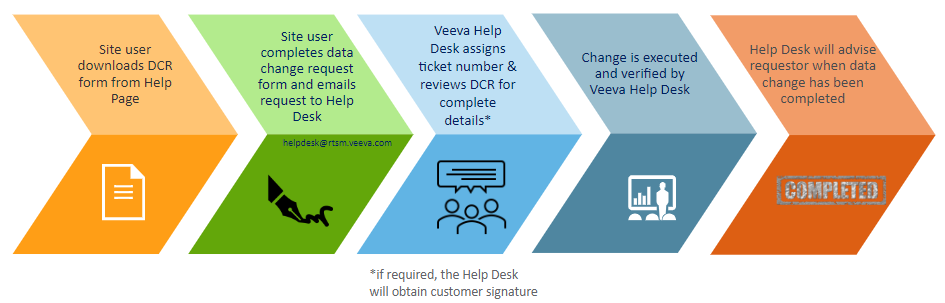
Data Change Request Form
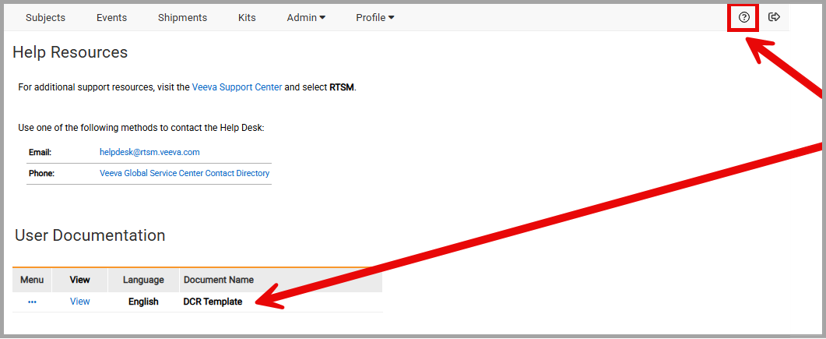
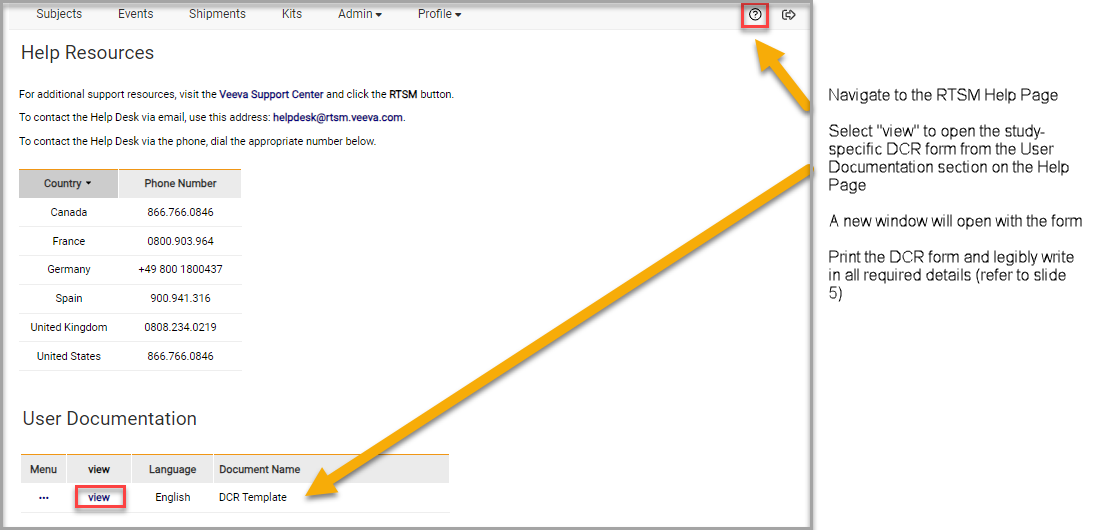
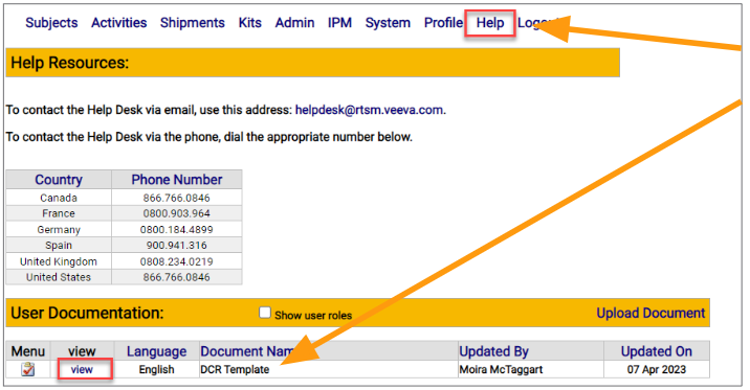
A Data Change Request form is located on the Help page of Veeva RTSM.
- Navigate to the RTSM Help Page
- Select View to open the study-specific DCR form from the User Documentation section on the Help Page
- A new window will open with the form
- Print the DCR form and legibly write in all required details
System Versions post 25r2
System Versions post 23r1
System Version 1-4
DCR Approval Requirements
Veeva and the Sponsor/CRO have indicated on the DCR form the types of changes that require Sponsor Approval.
If a signature is required, the change is not run until Veeva collects the customer signature.

NOTE: Data change types and approval requirements vary by study.
Filling out the DCR Form

Once the form is filled out completely, email the form to rtsmdcr@veeva.com