When site users log into Clinical Vault, the Site Connect > Site Home tab is displayed. This tab contains the following sections and information in the left navigation panel.
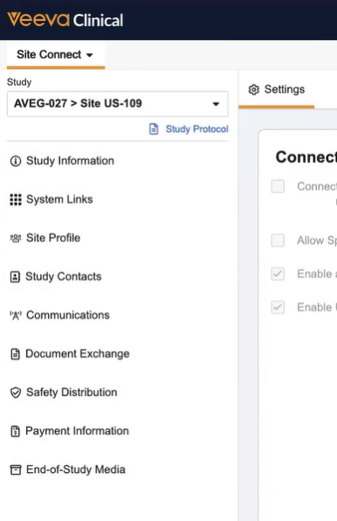
Study Selector
You can use the Study Selector to choose which Study you wish to view. Only Studies that the Sponsor/CRO has added to Site Connect will appear in the Study Selector.
Study Information
The Study Information section includes basic study information such as study name, phase, masking specifics, and product details. This section is not customizable.
System Links
The System Links section provides quick access to study applications with a central list of key links from your sponsor or CRO.
Site Profile
The Site Profile section is where you can manage your site staff accounts and site addresses.
Study Contacts
The Study Contacts section contains names, roles, and contact information for Sponsor and CRO study personnel.
Communications
The Communications section allows Sponsors to post study announcements, such as news and trial deadlines, in Site Connect. These posts are visible to Site users and are also sent to Sites as email notifications.
Document Exchange
The Document Exchange section displays the documents associated with the selected Study. From this section, you can view and take actions against documents.
Safety Distribution
The Safety Distribution section displays safety documents related to the selected Study. From this section, you can receive and review safety documentation and mark safety documents as Read.
Payment Information
The Payment Information section allows you to expedite payment receipt and reconciliation with the ability to see reimbursement data as it is tracked by the sponsor or CRO.
End of Study Media
The End-of-Study Media section contains all completed CRF documents that are sent by a Sponsor. Similar to the Document Exchange section, you can view and take actions against these documents, such as filing them to your connected SiteVault.
Site Settings
The Site Settings section allows you to select specific options for your Site Connect experience.
Attention Needed
If an orange dot appears on the bell (top, right) or on a menu item, this is an indicator that an item requires attention.